Cellular Respiration Resources

Overview
Plants and animals must take in and transform energy for use by cells. Plants, through photosynthesis, absorb light energy and form organic molecules such as glucose. Glucose has potential energy in the form of chemical energy stored in its bonds. This chapter covers the metabolic pathways of cellular respiration and describes the chemical reactions that use energy in glucose and other organic molecules to form adenosine triphosphate (ATP). ATP is the cell’s “energy currency” fueling virtually all energy requiring processes. The chemical reactions of cellular respiration are a series of oxidation- reduction (redox) reactions that are divided into three stages: glycolysis, the citric acid cycle and oxidative phosphorylation.
Learning Objectives
BIOLOGY I and II TAGS
- Explain how chemical processes impact the cellular processes of life.
- Describe the process of energy transfer from its source (the sun) through biological systems.
- Explain how energy moves through an ecosystem.
- Describe the enzymatic basis of the mechanisms that living organisms use to harvest energy.
- Outline the overall organization of the chemical pathways involved in cellular respiration and fermentation and how these pathways accomplish the processing of energy.
- Explain how cell regulatory mechanisms ensure balance in living systems that interact continuously with their environments
Recommended Textbook Resources
Chapter 7: Cellular Respiration
OpenStax: Biology 2e
Chapter 7, Cellular Respiration. Cellular respiration describes a series of redox reactions that transfer energy in organic molecules to ATP. In the presence of oxygen, aerobic cellular respiration includes glycolysis, pyruvate oxidation, the citric acid cycle and oxidative phosphorylation. Anaerobic respiration and fermentation produce ATP without oxygen. Regulation of enzymes that catalyze these reactions, primarily through negative feedback, ensure a constant supply of ATP in cells.
Student Assessment Activities
Student Assessment Activities are below; instructors can also Download a Cellular Respiration Student Activities Word File or View the Cellular Respiration Student Activity Google Doc.
Project 1:
The ATP producing reactions of cellular respiration and fermentation are critical in providing an accessible source of energy for cellular function.
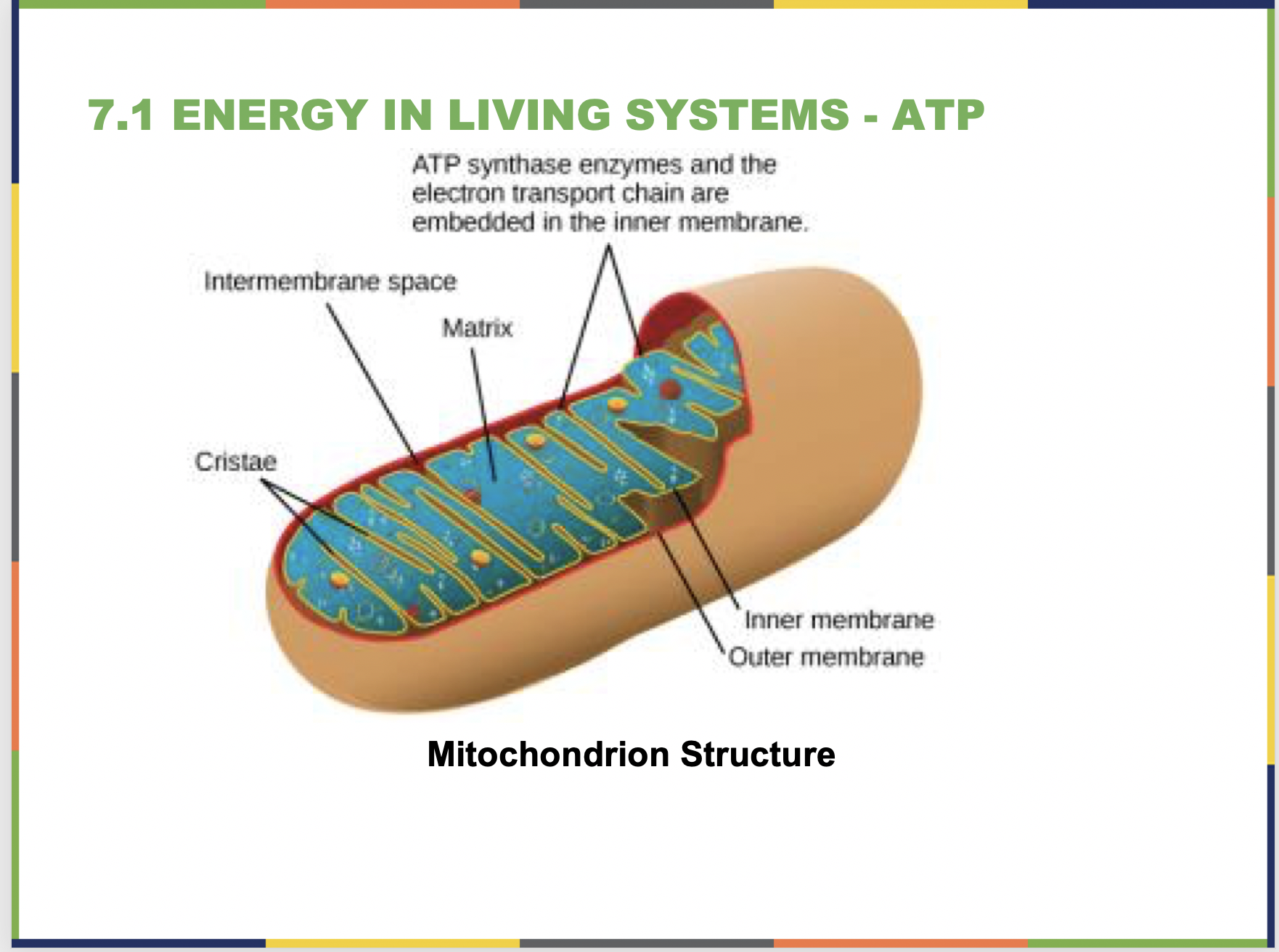
- Oxidative phosphorylation generates the majority of ATP in aerobic cellular respiration. Review the function of the electron transport chain (ETC) and chemiosmosis, then investigate the effect of dinitrophenol (DNP) on oxidative phosphorylation. Explain how DNP decreases ATP production through oxidative phosphorylation and how, though now banned, this molecule functioned as an effective weight-loss drug.
- Fermentation provides ATP when oxygen levels are too low for aerobic cellular respiration. Review the reactions of glycolysis, then investigate how tremetol can impact the metabolic pathways associated with lactic acid fermentation. Explain the symptoms associated with exposure to tremetol and why exercise can worsen symptoms associated with this metabolic poison.
Project 2:
Review the enzymes that catalyze each reaction of glycolysis. Note the reactions with enzymes that have names that include “kinase”. What do all these reactions have in common? Which reaction in the citric acid cycle would you predict is catalyzed by succinate thiokinase (also known as succinyl Co-A synthetase or succinyl Co-A ligase)? Explain the basic function of kinase enzymes.
Project 3:
Review section 7.1, Energy in Living Systems, with a focus on Electrons and Energy then answer the following questions. (1) Explain the relationship between oxidation/reduction and potential energy. (2) Explain the role of electron carriers in cellular respiration. (3) Identify which molecules are oxidized and which are reduced in the overall reaction for aerobic cellular respiration.
C6H12O6 + 6 O2 🡪 6 CO2 + 6 H2O
(4) Methanogens are prokaryotes from Domain Archaea that conduct anaerobic cellular respiration. Identify which molecules are oxidized and which are reduced in the overall reaction for methanogenesis.
CO2 + 4 H2 🡪 CH4 + 2 H2O